Precise real-world data. Conclusive real-world evidence.
Enhance your research with precise data and expert insights – supporting studies across industries.

OVERVIEW
Accelerate your real-world evidence studies
4.5k+
Peer-reviewed publications rely on MarketScan. Explore our research and success stories on the latest healthcare trends.
You bring the research questions, MarketScan will bring the answers.
Research grade data
Gain the granularity of fully-adjudicated closed claims data from payers across the US with actual cost and extensive longitudinal patient-views.
Streamlined cloud tooling
Enhance team efficiency and collaboration by leveraging a no-code application and a scalable cloud environment on Snowflake.
Expert research and analytics services
Draw insights and guidance from experts with deep experience in epidemiology, regulatory approvals, and health economics outcomes research (HEOR).
“We know that MarketScan data is trusted and of top quality. The real-world data has helped answer questions earlier, and for us, that is priceless because we are able to help our customers quicker and more efficiently.”
Paul Petraro, Global Head of Real World Evidence, Boehringer Ingelheim
“The questions that we ask with this type of data, you really can’t do anywhere else.”
Division Chief, Biomedical Informatics & Internal Medicine, Large University
“The longitudinal MarketScan data enabled us to follow patients for 5 years, demonstrating potentially long-term consequences that weren’t previously identified.”
Division Chief, Biomedical Informatics & Internal Medicine, Large University
“Because we now have access to such a large amount of clean data and we’re confident in the variables, we are hopeful that identifying fetal toxicities will go from ten years to less than five.”
Anick Bérard, PhD, FCAHS, FISPE, Senior Scientist, CHU Sainte Justine
CORE FUNCTIONALITY
Optimize outcomes with decisive real-world evidence
-
Employer-sourced
-
Actual costs
-
Longitudinal patient-view
-
Complete + representative
Regulatory submissions
Our large and fully adjudicated closed claims data enables scientifically robust reporting to regulatory bodies.
HEOR
Our experts connect actual cost to treatment patterns and health outcomes, helping you identify gaps and demonstrate value.
Epidemiology research
Our complete, representative data empowers researchers to explore burden of illness and treatments, minimizing bias and increasing confidence.

WHO WE HELP
Real-world data that fuels discovery across healthcare
Life sciences
Payers
Academic institutions
Government agencies
One source for data, expertise, and evidence generation
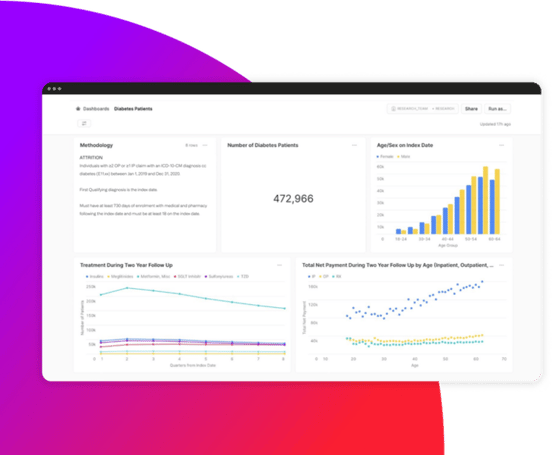
Gain immediate access to data with confidence
Analyze large volumes of data with ease

Uncover stronger insights with expert support and advanced linking
RESOURCES
Dig deeper into real-world evidence solutions
Learn about our relationship with Veradigm
We collaborated with Veradigm to bring a groundbreaking new database to healthcare research: the Linked Claims + EHR Database.
Insights Uncovered: A Real-World Data Podcast
MarketScan experts and series guests dive deep into the challenges and opportunities of using real-world data in healthcare and life sciences.
Unlock value with MarketScan’s advanced data linking
Experts from MarketScan and Biodesix discussed how advanced data linking drives deeper understanding, better decision‑making, and measurable organizational value.
